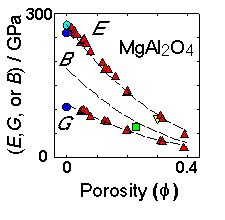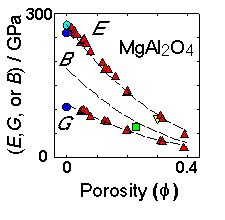The distribution of dopant in glass ceramics may be totally different due to the existence of glass phase.
Properties of yttrium oxide ceramics.
Results indicated that a higher sintering temperature benefited the preparation of ceramic cores with excellent properties.
Yttria ceramics have been widely investigated and used due to their significant physical and chemical properties such as high melting temperature 2430 c low thermal expansion high thermal.
It is an air stable white solid substance.
The cubic structure of yttrium oxide is stable to 1800 c.
These cases are illustrated by yttrium barium copper oxide ybco.
Densification aids are yttrium oxide plus aluminum oxide.
The thermal conductivity of yttrium oxide is 27 w m k.
This study examines the distribution of y 3 ions in a zro 2 sio 2 glass ceramic.
Yttria overview yttria yttrium oxide y 2 o 3 is a rare earth ceramic which exhibits both exceptional plasma resistance and an aptitude for high temperature applications.
The influences of yttrium oxide content and sintering temperature on properties of ceramic cores were studied carefully.
Two samples were prepared by hot pressing yttrium oxide doped and undoped 65 mol zro 2 35 mol sio 2 nanocrystalline.
In severely anisotropic materials there can be great variation of properties.
Densification occurs above 2100ºc by a liquid phase reaction and results in a glassy second phase.
As the temperature was above 1400 c the reaction of al 2 o 3 and yttrium oxide occurred leading to the formation of yag phases.
Ybco is a superconducting ceramic.
Was observed on heating the oxide to 1800 c.
The mechanical properties are generally superior to ssic but the high temperature properties and the corrosion resistance are not as good.
A change in lattice parameter of less than 0 007 a u.
That is it loses all resistance to electric current at extremely low temperatures.
Properties of yttrium oxide ceramics properties of yttrium oxide ceramics curtis c.
In air as indicated by petrographic x ray and differential thermal analyses.
Its structure consists of three cubes with yttrium or barium at the centre copper at the corners and oxygen at the middle of each edge with the exception of the middle cube.
The mean specific heat of y 2 o 3 to 1600 c.
Yttrium oxide also known as yttria is y 2 o 3.